Nonlinear Spectroscopy and Biophysical Chemistry
The central objective of our group is to use laser based nonlinear spectroscopy to probe physical properties in solution. In the area of molecular nonlinear optics our aim has been to exploit second harmonic light scattering (SHLS) as a probe for bulk physical properties of molecules in solution. Intensity of second harmonic scattered light is proportional to the molecular hyperpolarizability (β) which changes with a physical process as well as the symmetry of the molecular species in solution. Application of second harmonic light scattering in chemistry problems remains a very active area in our group.
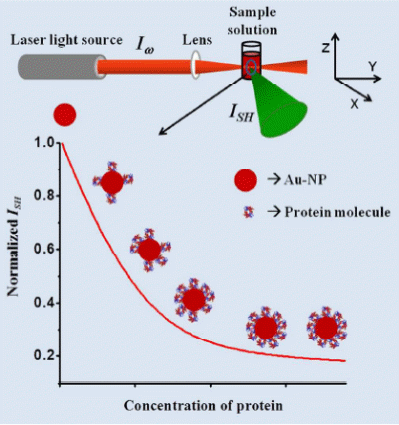
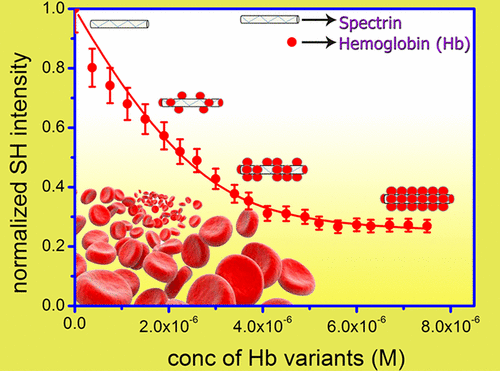
Our current interest is in the area of biophysical chemistry where we are investigating protein-protein (Fig.1) and nanoparticle-protein (Fig.2) interactions by SHLS in solution. Gold nanoparticles show huge SHLS signal due to large local surface polarization. Binding an analyte such as a protein, peptide, amino acid, etc. changes the surface polarization and consequently the second harmonic response. This change in SHLS signal upon adsorption of analytes on gold nanoparticle surface has been fitted to various known adsorption models to get the free energy of adsorption (ΔG) and equilibrium binding constant (K) of the analyte to the surface. With the rapid rise in interest in finding use of nanoparticles in various biomedical applications, characterization of protein-nanoparticle interaction has become increasingly important. This is mainly because proteins are always present in biological environment and the nanoparticles will have to come in contact with them in case they are used in cellular applications. The SHLS experiments are carried out at nanomolar or sub-nanomolar concentrations where protein aggregation is completely avoided and we make clean measurements. By doing temperature dependent SHLS experiments we get the enthalpy (ΔH) and entropy (ΔS) changes in the adsorption process. These data are necessary for designing nanoparticle-protein conjugates for diagnostic as well as therapeutic purposes and for benchmarking against molecular dynamics simulations of binding kinetics in solution. In protein-protein interaction we have looked at binding of hemoglobin variants with cytoskeletal protein spectrin and by SHLS experiments we were able to pick up differences in binding between two proteins with single mutation differences. The sensitivity of this technique allows us to apply this to a variety of problems where measurement techniques at low concentrations are lacking.
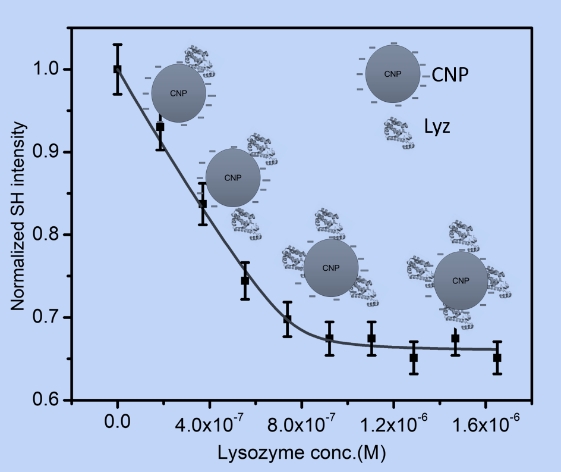
While fitting the adsorption data we have found both monolayer and multilayer adsorptions of proteins on the gold nanoparticle surface. A modified Langmuir type adsorption isotherm fitted the experimental data very well for monolayer formation processes. However, for multilayer formation, Brunauer, Emmett, and Teller (BET) model was necessary to fir the data. Protein adsorption on carbonaceous nanoparticles (CNPs) is found to be similar to that on GNPs (Fig.3) although the nature of the two surfaces are qualitatively different along with the mechanism of binding.
Representative Publications
-
Probing Protein Adsorption on Nanoparticle Surface by Second Harmonic Light Scattering.A. Das, A. Chakrabarti, and Puspendu Kumar DasPCCP, 18, 24325- 31 (2016)
-
Protein-protein Interaction Probed by Label-free Second Harmonic Light Scattering: Hemoglobin Adsorption on Spectrin Surface as a Case Study.Kamini Mishra, Abhijit Chakrabarti and Puspendu K. DasJ. Phys. Chem. B 121, 7797-7802 (2017)
-
Thermodynamics of adsorption of alcohol dehydrogenase on gold nanoparticle surface: A model based analysis versus direct measurement.Akriti Mishra, Kamini Mishra, Dipayan Bose, Abhijit Chakrabarti, and Puspendu K. DasPCCP, 23, 24365 - 24376 (2021)
-
Thermodynamics of multilayer protein adsorption on a gold nanoparticle surface.Akriti Mishra and Puspendu Kumar DasPCCP, 24, 22464-476 (2022)
-
Lysozyme Adsorption on Carbonaceous Nanoparticles Probed by Second Harmonic Light Scattering.Sourav Saikia, Jyoti Dutta, Akriti Mishra, and Puspendu Kumar DasPCCP (accepted, 2023)

